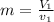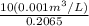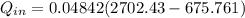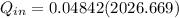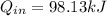Engineering, 15.02.2020 05:24 asyamelissa01
A rigid 14-L vessel initially contains a mixture of liquid water and vapor at 100°C with 12.3 percent quality. The mixture is then heated until its temperature is 180°C. The final state is superheated water and the internal energy at this state should be obtained by interpolation. Calculate the heat transfer required for this process. Use data from the steam tables.

Answers: 1


Other questions on the subject: Engineering



Engineering, 04.07.2019 18:20, maustin5323
Athin walled concentric tube exchanger is used to cool engine oil from 160°c to 60°c with water that is available at 25°c acting as a coolant. the oil and water flow rates are each at 2 kg/s, and the diameter of the inner tube is 0.5 m and the corresponding value of the overall heat transfer coefficient is 250 w/m2. oc. how long must the heat exchanger be to accomplish the desired cooling? cpwater=4.187 kj/kg-candcpengine el=2.035 kj/kg·°c, oil . 120]
Answers: 1

Engineering, 04.07.2019 18:20, cxttiemsp021
Atank with constant volume contains 2.27 kg of a mixture of water phases (liquid-vapor). in the initial state the temperature and the quality are 127 °c and 0.6, respectively. the mixture is heated until the temperature of 160 oc is reached. illustrate the process in a t-v diagram. then, determine (1) the mass of the vapor in kg at the initial state, (2) the final pressure in kpa.
Answers: 3
You know the right answer?
A rigid 14-L vessel initially contains a mixture of liquid water and vapor at 100°C with 12.3 percen...
Questions in other subjects:

History, 09.07.2019 02:30




Mathematics, 09.07.2019 02:30

Mathematics, 09.07.2019 02:30

Mathematics, 09.07.2019 02:30


Mathematics, 09.07.2019 02:30

Mathematics, 09.07.2019 02:30

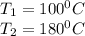
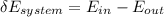
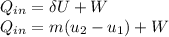
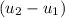 = corresponding change in the internal energy at state point 2 and 1
= corresponding change in the internal energy at state point 2 and 1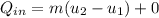
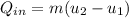
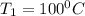 , the given quality of mixture of liquid water and vapor
, the given quality of mixture of liquid water and vapor 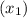 = 123% = 0.123
= 123% = 0.123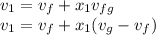
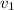 = specific volume at state 1
= specific volume at state 1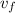 = specific volume of the liquid
= specific volume of the liquid 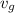 = specific volume of the liquid vapor mixture
= specific volume of the liquid vapor mixture 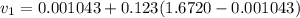
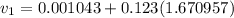
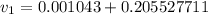
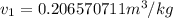
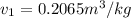
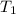 = 100°C and
= 100°C and 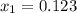 ;
;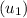 at the state 1
at the state 1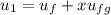
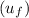 = 419.06 kJ/kg
= 419.06 kJ/kg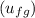 = 2087.0 kJ/kg
= 2087.0 kJ/kg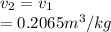
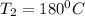 from the data in the steam tables
from the data in the steam tables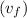 = 0.00113 m³/kg
= 0.00113 m³/kg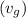 = 0.19384 m³/kg
= 0.19384 m³/kg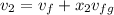
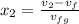
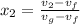
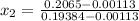
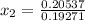
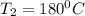
 = 1820.88 kJ/kg
= 1820.88 kJ/kg
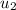 ≅ 2702.43 kJ/kg
≅ 2702.43 kJ/kg