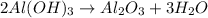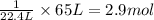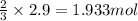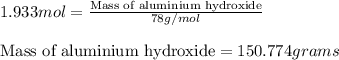Chemistry, 22.07.2019 16:00 navarreteanamen123
What mass of aluminium hydroxide is needed to decompose in order to produce 65.0 l of water at stp in stoichiometry?

Answers: 1


Other questions on the subject: Chemistry




Chemistry, 22.06.2019 18:50, christhegreat1
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1
You know the right answer?
What mass of aluminium hydroxide is needed to decompose in order to produce 65.0 l of water at stp i...
Questions in other subjects:


Mathematics, 31.01.2020 22:43


Advanced Placement (AP), 31.01.2020 22:43



Mathematics, 31.01.2020 22:43


Mathematics, 31.01.2020 22:43