
Chemistry, 29.07.2019 01:30 maddielr17
How much energy is required to decompose 800 g of pcl3, according to the reaction below? the molar mass of pcl3 is 137.32 g/mol and may be useful. 4 pcl3(g) → p4(s) + 6 cl2(g) δh∘rxn = +1207 kj how much energy is required to decompose 800 of , according to the reaction below? the molar mass of is 137.32 and may be useful. 4 (g) (s) + 6 (g) = +1207 1.76×103 kj 4.53×103 kj 2.31×103 kj 6.72×103 kj 5.90×103 kj?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:00, carvajalj2520
Explain what happens at the saturation point when adding salt to water at room temperature.
Answers: 1

Chemistry, 22.06.2019 14:00, emilyproce
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1

Chemistry, 23.06.2019 11:40, missmontgomery21
Which of the following observations indicates that an atom has neutrons? some uncharged particles are scattered by a beryllium atom when it hits a gold foil. some uncharged particles bounce back from a gold foil when it is bombarded with alpha particles. a radiation consisting of uncharged particles is emitted when alpha particles strike beryllium atoms. a radiation which attracts electrons is produced when a beryllium atom is bombarded with alpha particles.
Answers: 2
You know the right answer?
How much energy is required to decompose 800 g of pcl3, according to the reaction below? the molar...
Questions in other subjects:



Physics, 10.03.2020 03:05







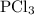 is
is  .
.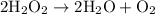
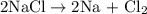
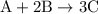
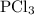 occurs as follows:
occurs as follows: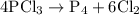
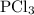 is as follows:
is as follows: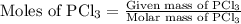 …… (1)
…… (1)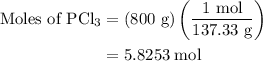
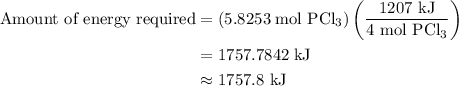
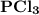 is 1757.8 kJ.
is 1757.8 kJ.

