19
Gold reacts with the elements in Group 7 of the periodic table.
0.175 g of gold reacts wit...

Chemistry, 24.06.2021 14:40 jsharma57p7enrw
19
Gold reacts with the elements in Group 7 of the periodic table.
0.175 g of gold reacts with chlorine.
The equation for the reaction is:
2 Au + 3 Cl2 - 2 AuCla
Calculate the mass of chlorine needed to react with 0.175 g of gold.
Give your answer in mg
Relative atomic masses (Ar): Cl = 35.5 Au = 197

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:50, emily9656
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3

Chemistry, 22.06.2019 19:10, asdfghhk9805
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2
You know the right answer?
Questions in other subjects:


Mathematics, 10.07.2019 03:00

Mathematics, 10.07.2019 03:00

Mathematics, 10.07.2019 03:00

Biology, 10.07.2019 03:00

Mathematics, 10.07.2019 03:00

Business, 10.07.2019 03:00

Mathematics, 10.07.2019 03:00


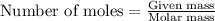 ......(1)
......(1)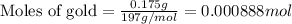
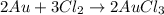
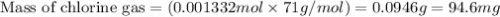 (Conversion factor: 1 g = 1000 mg)
(Conversion factor: 1 g = 1000 mg)

