
Chemistry, 24.05.2021 20:10 meganwintergirl
During a laboratory experiment, 36.12 grams of Aboz was formed when O2 reacted with aluminum metal at 280.0 K and 1.4 atm. What was the volume of O2
used during the experiment?
30 2+ 4A1 -- 2AI2O3
8.70 liters
9.22 liters
10.13 liters
12.81 liters

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:20, monsurviky
Amixture of gaseous sulfur dioxide and oxygen are added to a reaction vessel and heated to 1000 k where they react to form so3(g). if the vessel contains 0.669 atm so2(g), 0.395 atm o2(g), and 0.0851 atm so3(g) after the system has reached equilibrium, what is the equilibrium constant kp for the reaction: 2 so2(g) o2(g) ⇌ 2 so3(g)
Answers: 3


Chemistry, 22.06.2019 16:50, brandiwingard
What is conserved in the reaction shown below? h2(g) + cl2 (g) --> 2hcl(g)a. mass onlyb. mass and moles onlyc. mass, moles, and molecules onlyd. mass, moles, molecules, and volume
Answers: 2
You know the right answer?
During a laboratory experiment, 36.12 grams of Aboz was formed when O2 reacted with aluminum metal a...
Questions in other subjects:










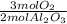 = 0.531 mol O₂
= 0.531 mol O₂

