
Chemistry, 21.05.2021 19:00 logan541972
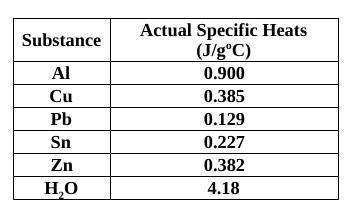
You had a 69.24 g piece of unknown metal and placed it into hot water at 103°C. You then performed the same experiment as done in class (specific heat of a metal lab) using a Styrofoam cup calorimeter (filled with 75.0 mL of water) and you found that the temperature of the water in the cup rose from 23.0˚C to 27.6˚C, what would the metal’s specific heat be? Identify the metal from the table. (The specific heat of water is 4.184 J/g) I need work to be shown

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, amandasantiago2001
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3

Chemistry, 22.06.2019 08:00, katelyn0579
Straightforward questions answered in the powerpoint slidesreaction: heating the starting materials under refluxwhat does it mean to heat under reflux? why do we choose water as the reflux solvent? what are boiling chips used for? why do we put a condenser on top of the reaction? why do we add heat and let the reaction stir for 30 minutes? why do we add sulfuric acid to the reaction after it cools as opposed to when it’s still hot? separation: filtration of precipitatewhy don’t we do an aqueous and organic extraction in the separatory funnel? why do you rinse the salicylic acid on the filter with ice cold water? purification: recrystallization of salicylic acid (no hot filtration needed)what is the difference in the amount of room temperature water vs. boiling water needed to dissolve the salicylic acid (assume a 1.2 gram yield of salicylic acid)? remember, in the lab if you need x ml of boiling water to dissolve a solid, then you should add a little more (definitely no more than 1.5 times the theoretical amount) to ensure it doesn’t recrystallize prematurely. analysis: melting point of salicylic acidwhat can you conclude if the melting point of the salicylic acid you just synthesized is 152-155oc and the 1: 1 mix of your product and “synthetic” salicylic acid is 151-154oc?
Answers: 1

Chemistry, 22.06.2019 12:00, carvajalj2520
Explain what happens at the saturation point when adding salt to water at room temperature.
Answers: 1

Chemistry, 23.06.2019 01:30, Nathaliasmiles
If a particle has z = 25 and 23 electrons, what is its charge?
Answers: 2
You know the right answer?
You had a 69.24 g piece of unknown metal and placed it into hot water at 103°C. You then performed t...
Questions in other subjects:



Mathematics, 19.03.2021 18:10





Mathematics, 19.03.2021 18:10

Mathematics, 19.03.2021 18:10



