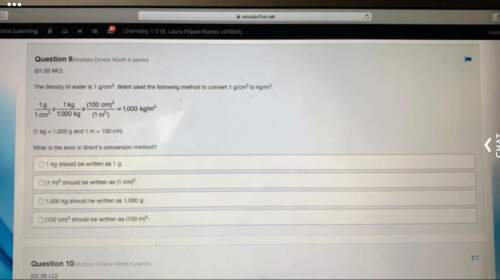
For each compound, circle the cation
element and underline the anion element.
u
=
...

Chemistry, 26.03.2021 19:20 claradendulk
For each compound, circle the cation
element and underline the anion element.
u
=
1. CrCl3
2. Hg 12
3. Mn_03
4. CuBr
5. Fe, o
6. Na2CO3
7. KNO
8. MgSO4
9. Mg(OH),
10. AIPO4
OD
5


Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:10, gonzalesalexiaouv1bg
Using complete sentences, explain how to predict the products and balance the reaction between sulfuric acid and potassium hydroxide.
Answers: 1

Chemistry, 22.06.2019 22:40, lindseyklewis1p56uvi
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization. a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution. part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 22.06.2019 23:10, ArielA13
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3
You know the right answer?
Questions in other subjects:


Mathematics, 06.05.2020 05:13

Mathematics, 06.05.2020 05:13

History, 06.05.2020 05:13

History, 06.05.2020 05:13

English, 06.05.2020 05:13

Mathematics, 06.05.2020 05:13

Mathematics, 06.05.2020 05:13

Mathematics, 06.05.2020 05:13

English, 06.05.2020 05:13