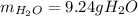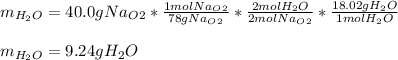A) What mass in grams of H20 is needed to react completely with 40.0 g of
Na2O2?
M(H2O)...

Chemistry, 26.02.2021 09:20 jagslovegirl
A) What mass in grams of H20 is needed to react completely with 40.0 g of
Na2O2?
M(H2O) =18.02g/mol
M(NA2O2)= 78g/mol
Ecuation:
2Na2O2 (s)+2h2O(I)—> 4NaOH(aq) + O2 (g)

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:00, kyllow5644
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3

Chemistry, 22.06.2019 19:50, VoidedAngel
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3

Chemistry, 23.06.2019 01:20, cedricevans41p4j3kx
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1
You know the right answer?
Questions in other subjects:

Mathematics, 21.03.2021 09:40

Mathematics, 21.03.2021 09:40

Mathematics, 21.03.2021 09:40


Biology, 21.03.2021 09:40

Mathematics, 21.03.2021 09:40

English, 21.03.2021 09:40

Mathematics, 21.03.2021 09:50

English, 21.03.2021 09:50

Advanced Placement (AP), 21.03.2021 09:50