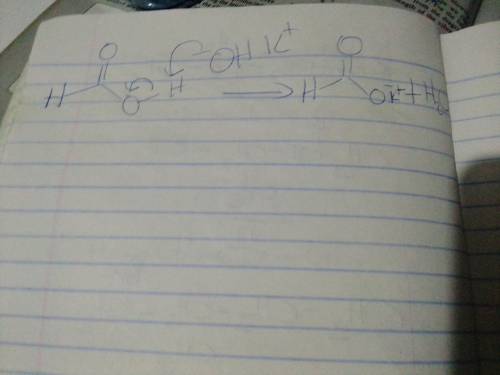Chemistry, 31.12.2020 19:00 sassycutie523
When formic acid is treated with potassium hydroxide (KOH), an acid-base reaction occurs, forming a carboxylate ion. For the mechanism, draw the curved arrows as needed. Include lone pairs and charges in your answer. Do not draw out any hydrogen explicitly in your products.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, maddietomlinson113
If the element whose electric configuration ends in the d sublevel, the element is calssified as? a. inner transition b. noble gases c. representative d. transition
Answers: 2

Chemistry, 22.06.2019 08:00, stephstewart1209
Which of the following observations indicates that there is a small, dense, positively charged part in the center of an atom? some uncharged particles are scattered by a gold foil. all uncharged particles are attracted towards a gold foil. all positively charged particles pass straight through a gold foil. some positively charged particles bounce back from a gold foil.
Answers: 2

Chemistry, 22.06.2019 10:10, andersonemma2222
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1
You know the right answer?
When formic acid is treated with potassium hydroxide (KOH), an acid-base reaction occurs, forming a...
Questions in other subjects:

Mathematics, 08.10.2019 02:40


English, 08.10.2019 02:40




English, 08.10.2019 02:40