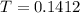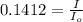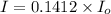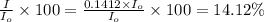Ireally need on this question!
the spectrophotometer really measures the percent of light t...

Chemistry, 30.01.2020 22:50 expeditionofsin
Ireally need on this question!
the spectrophotometer really measures the percent of light that is transmitted through the solution. the instrument then converts %t (transmittance) into absorbance by using the equation you determined in the pre-lab section. if the absorbance of a sample is 0.85, what is the percent light transmitted through the colored sample at this collected wavelength?
we only used the spectrophotometer to measure the absorbance and never got any %t values. so i have to convert absorbance to transmittance. how do i do this with the given absorbance of 0.85?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, cheyennemitchel238
Calculate the frequency of a wave in a spring toy. the wave has a speed of 1.1 meters per second and a wavelength of 0.1 meters. *
Answers: 2

Chemistry, 22.06.2019 19:50, jakaylathomas11
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3


Chemistry, 23.06.2019 08:00, codybrocs9624
Can anyone answer these questions? ? i need it before 1: 00pm today
Answers: 2
You know the right answer?
Questions in other subjects:



Mathematics, 05.05.2020 06:31

Mathematics, 05.05.2020 06:31



Mathematics, 05.05.2020 06:31



History, 05.05.2020 06:31

![A=\log \frac{I_o}{I}=-\log[T]](/tpl/images/0487/5067/baad9.png)
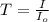
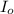 = incident light
= incident light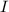 = transmitted light
= transmitted light![0.85=-\log[T]](/tpl/images/0487/5067/bf193.png)