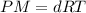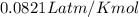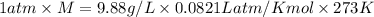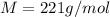Chemistry, 05.05.2020 06:31 rainbowsadie7140
A gaseous compound composed of sulfur and oxygen has a density of 9.88 g/L at stp. What is the molar mass of this gas?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, EMQPWE
In a spacecraft, the following reaction occurs: co2(g) + 2lioh(s) -> lico3(s) + h2o(i) (i attached picture of equation) how many liters of carbon dioxide will 4 moles of lithium hydroxide (lioh) absorb? (one mole of any gads occupies 22.4 l under certain conditions of temperature and pressure. assume those conditions for this equation.) 45l 6.0l 3.0l 34l
Answers: 1


Chemistry, 22.06.2019 12:30, skaterwolf1317
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2
You know the right answer?
A gaseous compound composed of sulfur and oxygen has a density of 9.88 g/L at stp. What is the molar...
Questions in other subjects:

Mathematics, 09.10.2019 09:00


Mathematics, 09.10.2019 09:00

Mathematics, 09.10.2019 09:00



Biology, 09.10.2019 09:00

Mathematics, 09.10.2019 09:00

History, 09.10.2019 09:00

Mathematics, 09.10.2019 09:00