Chemistry, 12.08.2020 06:01 maytce7237
A 100.0 mL sample of 0.10 M NH3 is titrated with 0.10 M HNO3. Determine the pH of the solution before the addition of any HNO3. The Kb of NH3 is 1.8 × 10-5.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:30, jonloya264
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 11:40, jerrysandoval22
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2

You know the right answer?
A 100.0 mL sample of 0.10 M NH3 is titrated with 0.10 M HNO3. Determine the pH of the solution befor...
Questions in other subjects:

Social Studies, 02.03.2020 19:50

History, 02.03.2020 19:50


Biology, 02.03.2020 19:50






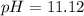
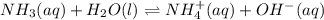
![Kb=\frac{[NH_4^+][OH^-]}{[NH_3]}](/tpl/images/0718/8402/ec4ce.png)
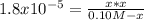
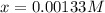
![pOH=-log([OH^-])=-log(0.00133)=2.88](/tpl/images/0718/8402/4fcb5.png)