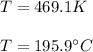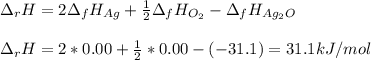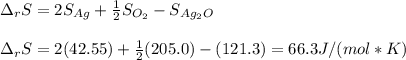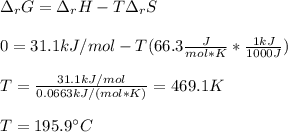Chemistry, 12.08.2020 05:01 jetjet123123
Given the data: Ag2O(s), = ‑31.1 kJ mol-1, S° = +121.3 J mol-1 K-1 Ag(s), = 0.00 kJ mol-1, S° = +42.55 J mol-1 K-1 O2(g), = 0.00 kJ mol-1, S° = +205.0 J mol-1 K-1 Calculate the temperature at which = 0 for the reaction, Ag2O(s) → 2 Ag(s) + ½ O2(g). Assume that, since the physical states do not change, and are independent of temperature between ‑50.0 °C and 950.0 °C.

Answers: 3


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 22:30, SavageKidKobe
Rank the four gases (air, exhaled air, gas produced from from decomposition of h2o2, gas from decomposition of nahco3) in order of increasing concentration of co2
Answers: 1

Chemistry, 23.06.2019 09:30, Dariana123
Which of the following is not a characteristic of a hydrogen bond? 1. it is responsible for the unusual physical properties of water. 2. it is weaker than a covalent bond. 3. it is stronger than other dipole-dipole interactions. 4. it can occur when hydrogen is covalently bound to very electronegative elements liks f, cl, br and i.
Answers: 1
You know the right answer?
Given the data: Ag2O(s), = ‑31.1 kJ mol-1, S° = +121.3 J mol-1 K-1 Ag(s), = 0.00 kJ mol-1, S° = +42....
Questions in other subjects:

Mathematics, 20.03.2020 00:06