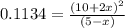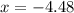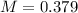Chemistry, 29.07.2020 04:01 leilaneedshelp3395
Consider the equilibrium system: N2O4 (g) = 2 NO2 (g) for which the Kp = 0.1134 at 25 C and deltaH rx is 58.03 kJ/mol. Assume that 1 mole of N2O4 and 2 moles of NO2 are introduced into a 5 L contains. What will be the equilibrium value of [N204]?
A) 0.358 M
B) 0.042 M
C) 0.0822 M
D) 0.928 M
E) 0.379 M

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, Falconpride4079
Construct the hypothetical phase diagram for metals a and b between room temperature (20c) and 700c, given the following information: * the melting temperature of metal a is 480c. • the maximum solubility of b in a is 4 wt% b, which occurs at 420c. • the solubility of b in a at room temperature is 0 wt% b. • one eutectic occurs at 420c and 18 wt% b–82 wt% a. • a second eutectic occurs at 475c and 42 wt% b–58 wt% a. • the intermetallic compound ab exists at a composition of 30 wt% b–70 wt% a, and melts congruently at 525c.• the melting temperature of metal b is 600c. • the maximum solubility of a in b is 13 wt% a, which occurs at 475c. • the solubility of a in b at room temperature is 3 wt% a.
Answers: 1

Chemistry, 22.06.2019 16:00, annsmith66
What statement goes against the kinetic theory of gases
Answers: 1

Chemistry, 23.06.2019 00:30, terryg4397
Fred is studying a substance that is made out of only one element. this means that
Answers: 1

Chemistry, 23.06.2019 01:30, emfranco1
Which of the following statements is true about energy quantization at the atomic level? electrons in the outermost orbits are the most stable. electrons in all the orbits around the nucleus have the same amount of energy. electrons in the orbit closest to the nucleus have the least amount of energy. electrons absorb or release the same amount of energy independent of the energy levels.
Answers: 1
You know the right answer?
Consider the equilibrium system: N2O4 (g) = 2 NO2 (g) for which the Kp = 0.1134 at 25 C and deltaH r...
Questions in other subjects:


Mathematics, 21.08.2020 20:01

Mathematics, 21.08.2020 20:01



Mathematics, 21.08.2020 20:01

Mathematics, 21.08.2020 20:01



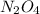 is 0.379 M
is 0.379 M
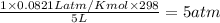
 =
= 

![K_p=\frac{[p_NO_2]^2}{[p_N_2O_4]}](/tpl/images/0714/6163/a9e41.png)