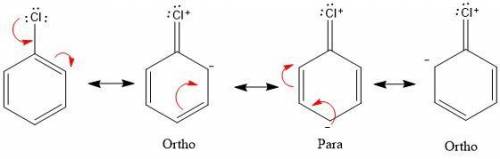
Chemistry, 04.07.2020 14:01 bumpydumper
In electrophilic aromatic substitution reactions, a chlorine substituent is . In electrophilic aromatic substitution reactions, a chlorine substituent is . an activator and a meta-director a deactivator and an ortho, para-director an activator and an ortho, para-director a deactivator and a meta-director none of the above

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 22:30, erinxmeow8
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons, neutrons, electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 22.06.2019 06:30, yolo123321
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

You know the right answer?
In electrophilic aromatic substitution reactions, a chlorine substituent is . In electrophilic aroma...
Questions in other subjects:










Mathematics, 20.11.2019 21:31