
Chemistry, 01.07.2020 22:01 corinnerodriguez2001
A ground state hydrogen atom absorbs a photon of light having a wavelength of 92.57 nm. It then gives off a photon having a wavelength of 954.3 nm. What is the final state of the hydrogen atom? Values for physical constants can be found in g

Answers: 3


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 18:50, emily9656
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3

Chemistry, 23.06.2019 01:00, tjeffers90028
Iron (fe) reacts with copper sulfate (cuso4) to form iron (ii) sulfate. in this reaction, cu2+ gains electrons to form cu. which statement is true about this reaction? fe(s) + cuso4(aq) → feso4(aq) + cu(s)
Answers: 3
You know the right answer?
A ground state hydrogen atom absorbs a photon of light having a wavelength of 92.57 nm. It then give...
Questions in other subjects:




Mathematics, 23.10.2019 23:00




Mathematics, 23.10.2019 23:00


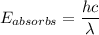
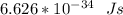
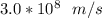
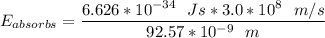
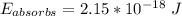
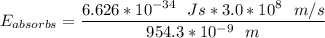
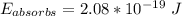
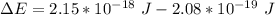
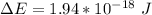
![\Delta E = R_H [\dfrac{1}{1^2}-\dfrac{1}{n^2_f}]](/tpl/images/0699/3893/e24b6.png)
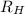 = Rydberg constant = 2.18 × 10⁻¹⁸ J
= Rydberg constant = 2.18 × 10⁻¹⁸ J![\dfrac{\Delta E}{R_H} = [\dfrac{1}{1^2}-\dfrac{1}{n^2_f}] \\ \\ \\ \dfrac{1.94*10^{-18} \ J}{2.18*10^{-18} \ J } = [\dfrac{1}{1^2}-\dfrac{1}{n^2_f}] \\ \\ \\ 0.889 = [\dfrac{1}{1^2}-\dfrac{1}{n^2_f}] \\ \\ \\ 1 - 0.889 = \dfrac{1}{n^2_f} \\ \\ \\ 0.111= \dfrac{1}{n^2_f} \\ \\ \\ {n^2_f} = \dfrac{1}{0.111} \\ \\ \\ {n^2_f} = 9 \\ \\ \\ {n_f} = \sqrt{9} \\ \\ \\ \mathbf{n_f = 3}](/tpl/images/0699/3893/e93b8.png)



