
Chemistry, 11.06.2020 01:57 herringalyssa
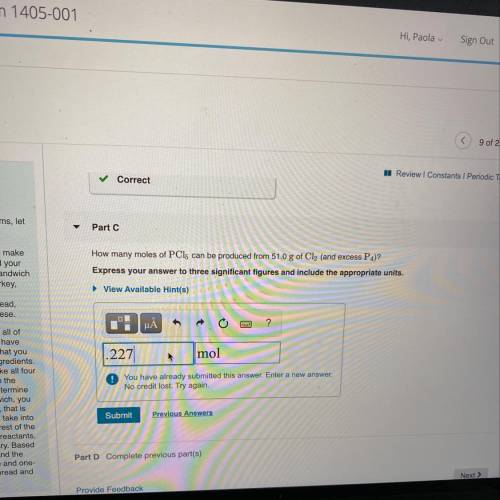
How many moles of PC15 can be produced from 51.0 g of Cl2 (and excess P4)?
Express your answer to three significant figures and include the appropriate units.
LIT.ITS NOT .227 or .228

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:00, fgcherubin
Consider the balanced equation below. n2h4 + 2h2o2 n2 + 4h2o what are the mole ratios of hydrazine (n2h4) to hydrogen peroxide (h2o2) and hydrazine to water? 1: 2 and 1: 4 1: 3 and 1: 4 1: 2 and 3: 5 1: 3 and 3: 5
Answers: 3

Chemistry, 22.06.2019 05:40, yah2muchh
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2


Chemistry, 22.06.2019 11:00, bigwaYne
Imagine that twenty i. u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2
You know the right answer?
How many moles of PC15 can be produced from 51.0 g of Cl2 (and excess P4)?
Express your answer to t...
Questions in other subjects:

Mathematics, 19.05.2020 16:59

Mathematics, 19.05.2020 16:59

Mathematics, 19.05.2020 16:59

Mathematics, 19.05.2020 16:59








