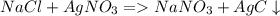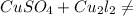Balance the following chemical reactions:
1. Pb(NO₃)₂ → PbO + NO₂ + O₂
2. CH₄...

Balance the following chemical reactions:
1. Pb(NO₃)₂ → PbO + NO₂ + O₂
2. CH₄ + O₂ → CO₂ + H₂O
3. Cu + AgNO₃ → Cu(NO₃)₂ + Ag
4. MnO₂ + HCl → MnCl₂ + H₂O + Cl₂
5. Pb(NO₃)₂ + NaCl → PbCl₂ + NaNO₃
Write the chemical reaction and balance it.
1. A solution of potassium chloride when mixed with silver nitrate solution, an insoluble
white substance is formed.
2. Copper sulphate on treatment with potassium iodide precipitates cuprous iodide(Cu₂l₂),
liberates iodine gas and also forms potassium sulphate.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:40, deedee363
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 11:00, jodonw5616
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1


Chemistry, 22.06.2019 23:00, catdog5225
What is formed when amino acids form long chains or polymerize
Answers: 1
You know the right answer?
Questions in other subjects: