
Chemistry, 08.04.2020 06:40 cancerbaby209
When 1 mol of methane is burned at constant pressure, −890 kJ/mol of energy is released as heat. If a 3.72 g sample of methane is burned at constant pressure, what will be the value of ∆H? (Hint: Convert the grams of methane to moles. Also make sure your answer has the correct sign for an exothermic process.) Answer in units of kJ.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, advancedgamin8458
Consider the nuclear equation below. 239 > x + 4 he 94 2 what is x? 1.235 cm 96 2.243 u 92 3.235 u 92 4.243 cm 96
Answers: 2

Chemistry, 22.06.2019 20:30, allofthosefruit
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3

Chemistry, 22.06.2019 21:30, djdjdjdbdbjx
What is another way to determine mass times acceleration?
Answers: 1

Chemistry, 22.06.2019 23:30, lizdeleon248
The sum of the oxidation numbers in a neutral compound is always
Answers: 2
You know the right answer?
When 1 mol of methane is burned at constant pressure, −890 kJ/mol of energy is released as heat. If...
Questions in other subjects:










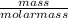
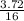 = 0.23moles
= 0.23moles 

