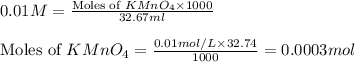Chemistry, 20.03.2020 10:46 jdisalle476
A sample of an Iron Oxalato complex salt weighting 0.13 grams requires 32.74 mL of 0.01 M KMnO4 to turn the solution a very light pink color at the quivalence point. Calculate the number of moles of KMnO4 added.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, Thomas7785
Two things that biomedical has invented or innovated
Answers: 1



Chemistry, 22.06.2019 03:00, HHHHHHHHHMMMMMMMMM
About 70 percent of the earth's surface is water-covered, and about 96.5 percent of all earth's water is salt water. identify the watery feature on earth that is made of freshwater rather than salt water. a) bay b) glacier c) ocean d) sea it is not incomplete this is the true question
Answers: 1
You know the right answer?
A sample of an Iron Oxalato complex salt weighting 0.13 grams requires 32.74 mL of 0.01 M KMnO4 to t...
Questions in other subjects:






Mathematics, 20.10.2019 13:10




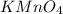 added are 0.0003
added are 0.0003 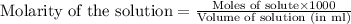 .....(1)
.....(1)