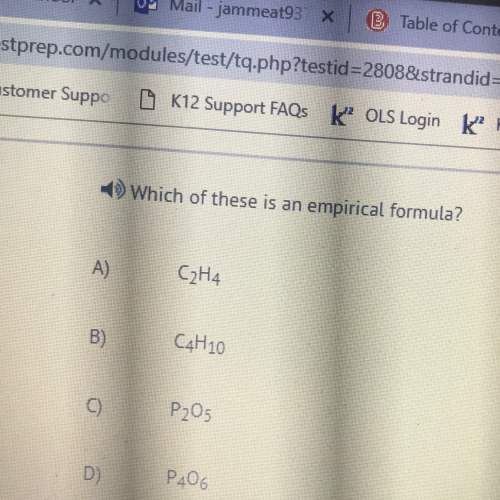
A 30.0-mL sample of 0.165 M propanoic acid is titrated with 0.300 MKOH.
A.) Calculate th...

Chemistry, 19.03.2020 09:03 diamond2738
A 30.0-mL sample of 0.165 M propanoic acid is titrated with 0.300 MKOH.
A.) Calculate the pH at 0 mL of added base.
Express your answer using two decimal places.
B.) Calculate the pH at 5 mL of added base.
C.) Calculate the pH at 10 mL of added base.
D.) Calculate the pH at the equivalence point.
Express your answer using two decimal places.
E.) Calculate the pH at one-half of the equivalence point.
Express your answer using two decimal places.
F.) Calculate the pH at 20 mL of added base.
Express your answer using two decimal places.
G.) Calculate the pH at 25 mL of added base.
Express your answer using two decimal place

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:00, XxrazorxX11
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1


Chemistry, 22.06.2019 12:30, UaRemomGAY
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

You know the right answer?
Questions in other subjects: