Chemistry, 13.03.2020 04:53 maevemboucher78
A mixture initially contains A, B, and C in the following concentrations: [A] = 0.650 M, [B] = 1.35 M, and [C] = 0.300 M. The following reaction occurs and equilibrium is established: A+2B<->C
At equilibrium, [A] = 0.550 M and [B] = 0.400 M. Calculate the value of the equilibrium constant, Kc

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:20, kingsqueen883
Consider the two electron arrangements for neutral atoms a and b. are atoms a and b the same element? a - 1s2, 2s2, 2p6, 3s1 b - 1s2, 2s2, 2p6, 5s1
Answers: 3

Chemistry, 21.06.2019 20:10, irene4523
Why is the vapor pressure of a warm lake higher than the vapor pressure of a cold lake? o a. warm water has a greater heat of vaporization. ob. warm water evaporates more quickly. cool water evaporates more quickly. od. cool water has a greater heat of vaporization.
Answers: 1

Chemistry, 22.06.2019 01:30, MickeyxX7096
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 09:00, Ezekielcassese
What is the percentage composition of carbon in the compound ch4
Answers: 1
You know the right answer?
A mixture initially contains A, B, and C in the following concentrations: [A] = 0.650 M, [B] = 1.35...
Questions in other subjects:

Mathematics, 05.07.2019 22:30



Mathematics, 05.07.2019 22:30

Mathematics, 05.07.2019 22:30



English, 05.07.2019 22:30


Mathematics, 05.07.2019 22:30

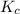 for given reaction is 0.465
for given reaction is 0.465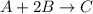
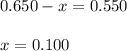
![K_c=\frac{[C]}{[A][B]^2}](/tpl/images/0546/2117/240ef.png)