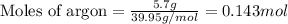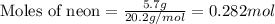Chemistry, 10.03.2020 07:22 lololol270
A gas mixture is made by combining 5.7 g each of Ar , Ne , and an unknown diatomic gas. At STP, the mixture occupies a volume of 12.88 L. What is the molar mass of the unknown gas?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, kylieweeks052704
Which of the following did jj thompson discover about atoms? a)an atom has an internal structure. b) atoms are tiny indivisible particles. c)electrons orbit the nucleus of an atom. d) the nucleus of an atom contains protons and neutrons.
Answers: 2

Chemistry, 22.06.2019 06:30, dpchill5232
Suppose a lab group reports a ppercent yield of sand of 105. is it really possible to collect more sand than was originally represented? what is the possible explanation for the extra product?
Answers: 2


Chemistry, 22.06.2019 21:30, MJyoungboy
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3
You know the right answer?
A gas mixture is made by combining 5.7 g each of Ar , Ne , and an unknown diatomic gas. At STP, the...
Questions in other subjects:






Chemistry, 09.04.2020 16:48



Mathematics, 09.04.2020 16:48

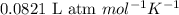
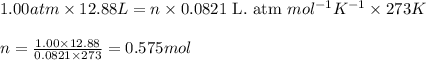
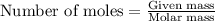 .....(1)
.....(1)