
Chemistry, 25.02.2020 06:22 Itsyourgirllulu
In the contact process, sulfuric acid is manufactured by first oxidizing SO2 to SO3, which is then reacted with water. The reaction of SO2 with O2 is
2SO2(g) 1 O2(g) m 2SO3(g)
A 2.000-L flask was filled with 0.0400 mol SO2 and 0.0200 mol O2. At equilibrium at 900 K, the flask contained 0.0296 mol SO3. How many moles of each substance were in the flask at equilibrium?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 23:00, ayoismeisalex
Matches the chemical name of each oxide of phosphorus to its chemical formula
Answers: 2


You know the right answer?
In the contact process, sulfuric acid is manufactured by first oxidizing SO2 to SO3, which is then r...
Questions in other subjects:

History, 27.09.2020 22:01








Advanced Placement (AP), 27.09.2020 22:01

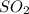 =0.0104
=0.0104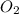 =0.0052
=0.0052

