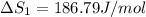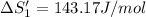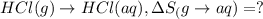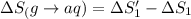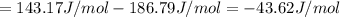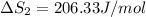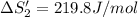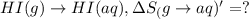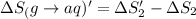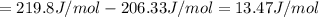All of the hydrogen halides (H-X) are gaseous in their natural state, but dissolve in water to form the aqueous phase.
Entropies (S) for the gaseous H-X molecules (before reaction) are:
HCl (g): 186.79 J/mol
HI (g): 206.33 J/mol
Entropies (S) for the H-X molecules dissolved/solvated in water (after reaction) are:
HCl (aq): 143.17 J/mol
HI (aq): 219.8 J/mol
1. Calculate the ΔS that each of these H-X compounds undergoes as it transitions from the gas phase to the aqueous phase.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:00, happy121906
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2



Chemistry, 23.06.2019 07:50, alexusnicole817
Asolution is produced in which water is the solvent and there are four solutes. which of the solutes can dissolve better if the solution is heated?
Answers: 1
You know the right answer?
All of the hydrogen halides (H-X) are gaseous in their natural state, but dissolve in water to form...
Questions in other subjects:

Mathematics, 06.05.2021 22:30


Mathematics, 06.05.2021 22:30







![\Delta S=[\text{Sum of entropy of products}]-[\text{Sum of entropy of reactants}]](/tpl/images/0519/3497/4a681.png)