
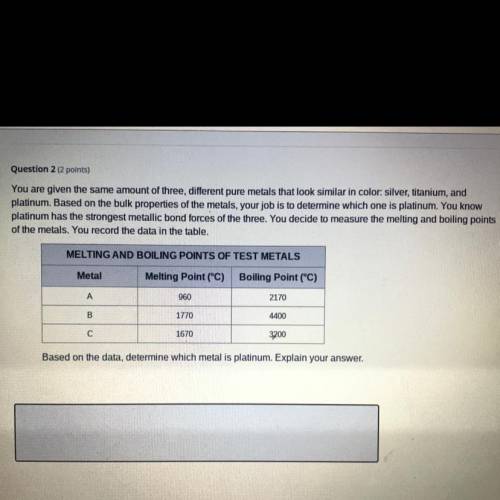
You are given the same amount of three different pure metals that look similar in color: silver, titanium, and platinum. Based on the bulk properties of the metals, your job is to determine which one is platinum. You know platinum has the strongest metallic bond forces of the three. You decide to measure the melting and boiling points of the metals. You record the data in the table.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, sotoamerica0814
98 ! and brainliest plz ! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 04:30, salvadorperez26
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 22.06.2019 10:10, babyphoraaaaa
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate, m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3
You know the right answer?
You are given the same amount of three different pure metals that look similar in color: silver, tit...
Questions in other subjects:

Mathematics, 24.02.2021 20:10


Mathematics, 24.02.2021 20:10

Mathematics, 24.02.2021 20:10


Health, 24.02.2021 20:10

English, 24.02.2021 20:10

History, 24.02.2021 20:10





