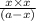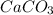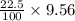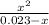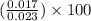Chemistry, 21.01.2020 01:31 pineapplepizaaaaa
The dissociation of calcium carbonate has an equilibrium constant of kp= 1.20 at 800°c. caco3(s) ⇋ cao(s) + co2(g)
a.) what is the kc for the reaction?
b.) if you place 20.0 g of caco3 in a 2.50 l container at 800°c, what is the pressure of co2 in the container?
c.) what percentage of the original 20.0 g sample of caco3 remains undecomposed at equilibrium?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 23.06.2019 00:00, ahmedeldyame
#20 which type of bond is formed when bases pair in dna? ionic bond covalent bond coordinate bond hydrogen bond
Answers: 1

Chemistry, 23.06.2019 01:20, hflores0001
How can parts of a solution be separated by chromatography?
Answers: 1

Chemistry, 23.06.2019 03:00, sharondacarruth1656
Is it safe to take 450mg of diphenhydramine hydrochloride?
Answers: 1
You know the right answer?
The dissociation of calcium carbonate has an equilibrium constant of kp= 1.20 at 800°c. caco3(s) ⇋ c...
Questions in other subjects:

Mathematics, 26.10.2020 20:50


English, 26.10.2020 20:50

Biology, 26.10.2020 20:50

Mathematics, 26.10.2020 20:50



Mathematics, 26.10.2020 20:50

Mathematics, 26.10.2020 20:50

Social Studies, 26.10.2020 20:50

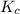 and
and 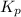 is as follows.
is as follows.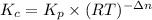
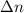 = 1
= 1
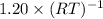
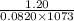
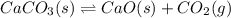
![p[CO_{2}] = K_{p}](/tpl/images/0463/2934/4d1a8.png) = 1.20
= 1.20
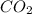 in the container is 1.20.
in the container is 1.20.![K_{c} = \frac{[CaO][CO_{2}]}{[CaCO_{3}]}](/tpl/images/0463/2934/031cb.png)