
Chemistry, 20.12.2019 23:31 andreamartinez7876
Use mathematical software or an electronic spreadsheet to examine thetime dependence of [i] in the reaction mechanism a →i→p (k1,k2). youmay either integrate eqn 22.39 numerically (seeappendix2) or use eqn 22.40directly. in all the following calculations, use [a]0=1 mol dm−3and a timerange of 0 to 5 s. (a) plot [i] against tfork1=10 s−1andk2=1 s−1. (b) increasethe ratio k2/k1steadily by decreasing the value of k1and examine the plot of [i]againsttat each turn. what approximation about d[i]/dtbecomesincreasingly valid?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 22:00, cooljariel11
Give more examples of this type of heat transfer:
Answers: 1


Chemistry, 23.06.2019 00:00, scottykinkade7860
Mercury turns to a vapor at 629.88 k. how much heat is lost when 75.0 g of mercury vapor at 650 k condenses to a liquid at 297 k?
Answers: 1

Chemistry, 23.06.2019 01:00, jaidencoolman2510
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2
You know the right answer?
Use mathematical software or an electronic spreadsheet to examine thetime dependence of [i] in the r...
Questions in other subjects:

Mathematics, 30.12.2019 23:31


Mathematics, 30.12.2019 23:31

Geography, 30.12.2019 23:31

Mathematics, 30.12.2019 23:31


Mathematics, 30.12.2019 23:31

Mathematics, 30.12.2019 23:31

Mathematics, 30.12.2019 23:31

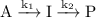
![\text{[I]} = \dfrac{k_{1}\text{[A]}_{0}}{k_{2} - k_{1}} \left(e^{-k_{1}t} - e^{-k_{2}t} \right )](/tpl/images/0428/3191/dc9ab.png)
![\text{[I]} = \dfrac{10}{-9} \left(e^{-10t} - e^{-t} \right )](/tpl/images/0428/3191/34abe.png)
![\mathbf{\dfrac{\text{d[I]}}{\text{d}t}=0}](/tpl/images/0428/3191/041dc.png)
![Use mathematical software or an electronic spreadsheet to examine thetime dependence of [i] in the r](/tpl/images/0428/3191/f5590.jpg)
![Use mathematical software or an electronic spreadsheet to examine thetime dependence of [i] in the r](/tpl/images/0428/3191/65bc4.jpg)



