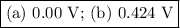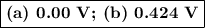Chemistry, 29.01.2020 07:50 SmokyWolves607
Calculate ℰ° values for the galvanic cells described below. (a) cr3+(aq) + cl2(g) equilibrium reaction arrow cr2o72-(aq) + cl -(aq) v (b) io3-(aq) + fe2+(aq) equilibrium reaction arrow fe3+(aq) + i2(aq)

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:20, Matseleng3775
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1

You know the right answer?
Calculate ℰ° values for the galvanic cells described below. (a) cr3+(aq) + cl2(g) equilibrium reacti...
Questions in other subjects:


Mathematics, 26.10.2019 01:43



Mathematics, 26.10.2019 01:43

English, 26.10.2019 01:43




Social Studies, 26.10.2019 01:43