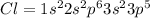Chemistry, 06.07.2019 15:00 cookiee835
Write the electron configuration for a chlorine atom. calculate the total number of electrons in each energy level and state which energy levels are not full. i know that the electron configuration for a chlorine atom is [ne] 3s2 3p5 but i need figuring out the total number of electrons in each energy level and state. : )

Answers: 1


Other questions on the subject: Chemistry




Chemistry, 23.06.2019 00:30, DragonLovely
•hydration •dissociation •dissolving which one goes to which
Answers: 1
You know the right answer?
Write the electron configuration for a chlorine atom. calculate the total number of electrons in eac...
Questions in other subjects:



Mathematics, 01.12.2021 22:10



Mathematics, 01.12.2021 22:10

Mathematics, 01.12.2021 22:10


Mathematics, 01.12.2021 22:10