
Suppose an oven's radiation wavelength is 0.125 m. a container with 350.00 g of water was placed in the oven, and the temperature of the water rose from 20.0°c to 80.0°c. how many photons of this microwave radiation were required to heat the water? (assume that all the energy from the radiation was used to raise the temperature of the water.) the specific heat of water is 4.18 j/g°c.

Answers: 1


Other questions on the subject: Physics

Physics, 21.06.2019 20:00, Tonilynnpinto63
What happens to atoms and chemical bonds during a reaction?
Answers: 1



Physics, 23.06.2019 01:30, theworld58
The dominant wavelength of energy emitted by the sun is
Answers: 2
You know the right answer?
Suppose an oven's radiation wavelength is 0.125 m. a container with 350.00 g of water was placed in...
Questions in other subjects:

Mathematics, 21.01.2021 19:10




Biology, 21.01.2021 19:10

English, 21.01.2021 19:10

Mathematics, 21.01.2021 19:10

Mathematics, 21.01.2021 19:10

Chemistry, 21.01.2021 19:10

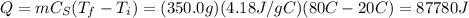
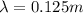 , so the energy of a single photon of this radiation is
, so the energy of a single photon of this radiation is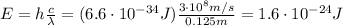
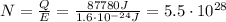 photons
photons


