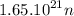Answers: 3


Other questions on the subject: Physics

Physics, 22.06.2019 12:40, ericsmith19
Afrequency generator sends a 550 hz sound wave through both water and ice. what is the difference in wavelength between the wave produced in ice and the wave produced in water?
Answers: 3

Physics, 22.06.2019 13:30, Bra1nPowers
6–48 bananas are to be cooled from 24 to 138c at a rate of 215 kg/h by a refrigeration system. the power input to the refrigerator is 1.4 kw. determine the rate of cooling, in kj/ min, and the cop of the refrigerator. the specific heat of banana above freezing is 3.35 kj/kg·8c.
Answers: 3

Physics, 22.06.2019 15:00, chynahgibsonwinfrey
What happens when a rubber rod is rubbed with a piece of fur?
Answers: 1

Physics, 22.06.2019 16:00, autumnstalvey67
Marvin records scientific data about lake superior. for which property has marvin forgotten to use an si unit of measurement? shore length high temperature low temperature annual precipitation
Answers: 3
You know the right answer?
Whats the gravitational force between mars and the sun...
Questions in other subjects:


Biology, 11.10.2019 00:00

History, 11.10.2019 00:00

Mathematics, 11.10.2019 00:00



Social Studies, 11.10.2019 00:00

Mathematics, 11.10.2019 00:00

Mathematics, 11.10.2019 00:00

Mathematics, 11.10.2019 00:00