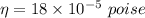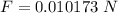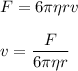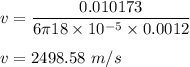Physics, 14.06.2021 14:20 isabellebisio
Calculate the terminal velocity of a rain drop of radius 0.12cm

Answers: 2


Other questions on the subject: Physics

Physics, 22.06.2019 00:10, oktacos
The energy released by a chemical reaction can be measured using a calorimeter. when barium hydroxide octahydrate crystals are reacted with dry ammonium chloride inside of a coffee cup calorimeter, the temperature of the 18.00 g of water in the calorimeter decreases from 30.0°c to 8.0°c. the equation for calculating energy absorbed or released by a reaction is: where q is the energy released or absorbed, m is the mass of water in the calorimeter, cp is the specific heat of water, and δt is the observed temperature change. if the specific heat of liquid water is 4.19 j/g·°c, how much energy was absorbed by the reaction?
Answers: 3

Physics, 22.06.2019 10:30, alwaysneedhelp8420
If y gets smaller as x gets bigger and y have an relationship?
Answers: 1

Physics, 22.06.2019 12:30, cardsqueen
Consider a hydrogen atom in the ground state. what is the energy of its electron? =e= jj now consider an excited‑state hydrogen atom. what is the energy of the electron in the =5n=5 level? =e5= j
Answers: 3

Physics, 22.06.2019 16:40, ashleypere99
Panel bc in fig. p2.76 is semi-circular, with the 3 meter radius and horizontal straight edge through point b. compute (a) the hydrostatic force of the water on the panel, (b) its center of pressure, and (c) the moment of this force about point b. assume atmospheric pressure on the dry side of the panel
Answers: 3
You know the right answer?
Calculate the terminal velocity of a rain drop of radius 0.12cm...
Questions in other subjects:

Mathematics, 19.08.2019 21:00

English, 19.08.2019 21:00



Social Studies, 19.08.2019 21:00


Mathematics, 19.08.2019 21:00

Physics, 19.08.2019 21:00


Geography, 19.08.2019 21:00