Answers: 1


Other questions on the subject: Physics

Physics, 22.06.2019 03:30, zahradawkins2007
As part of an industrial process, air as an ideal gas at 10 bar, 400k expands at steady state through a valve to a pressure of 4 bar. the mass flow rate of air is 0.5 kg/s. the air then passes through a heat exchanger where it is cooled to a temperature of 295k with negligible change in pressure. the valve can be modeled as a throttling process, and kinetic and potential energy effects can be neglected. (a) for a control volume enclosing the valve and heat exchanger and enough of the local surroundings that the heat transfer occurs at the ambient temperature of 295 k, determine the rate of entropy production, in kw/k. (b) if the expansion valve were replaced by an adiabatic turbine operating isentropically, what would be the entropy production? compare the results of parts (a) and (b) and discuss.
Answers: 3

Physics, 22.06.2019 05:30, GreenHerbz206
Could someone . i have tried to solve myself but my calculation is off
Answers: 3


Physics, 22.06.2019 06:30, bluevibe
2kg of refrigerant 134a undergoes a polytropic process in a piston-cylinder assembly from an initial state of saturated vapor at 2 bar to a final state of 12 bar, 80 degree c. a)determine the work for the process in kj. b)sketch the process on a p-v diagram.
Answers: 2
You know the right answer?
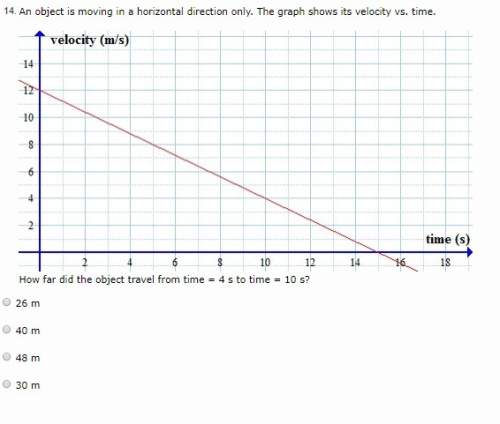
An object is moving in a horizontal direction only. the graph shows its velocity vs. time. (see grap...
Questions in other subjects:


Chemistry, 24.11.2020 23:20


Chemistry, 24.11.2020 23:20

Mathematics, 24.11.2020 23:20

Biology, 24.11.2020 23:20

Spanish, 24.11.2020 23:20