
Physics, 14.04.2021 01:10 FriesAintHere1392
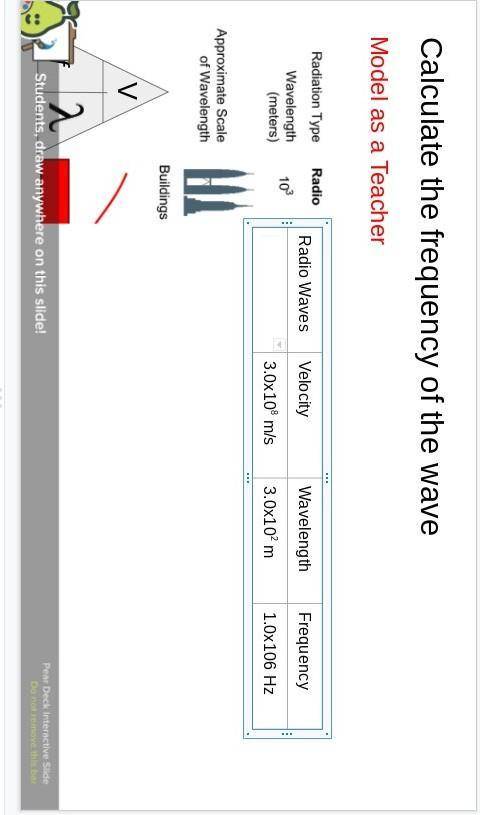
Someone please help I have no idea what to do

Answers: 3


Other questions on the subject: Physics


Physics, 22.06.2019 07:50, dimondqueen511
Calculate the ratio of h+ ions to oh– ions at a ph = 6. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 6. are they the same? why or why not? record your explanation in table a. what is the concentration of h+ ions at a ph = 6? mol/l what is the concentration of oh– ions at a ph = 6? mol/l what is the ratio of h+ ions to oh– ions at a ph = 6? : 1
Answers: 1

Physics, 22.06.2019 10:50, afonseca73
Asheet of steel 1.5 mm thick has nitrogen atmospheres on both sides at 1200oc and is permitted to achieve a steady-state diffusion condition. the diffusion coefficient for nitrogen in steel at this temperature is 6 x 10- 11 m2 /s, and the diffusion flux is found to be 1.2 x 10-7 kg/m2 -s. also, it is known that the concentration of nitrogen in the steel at the high-pressure surface is 4 kg/m3 . how far into the sheet from this high-pressure side will the concentration be 2.0 kg/m3 ? assume a linear concentration profile.
Answers: 3
You know the right answer?
Someone please help I have no idea what to do
...
...
Questions in other subjects:

Mathematics, 18.07.2019 01:30

History, 18.07.2019 01:30

History, 18.07.2019 01:30

Mathematics, 18.07.2019 01:30

Arts, 18.07.2019 01:30

Computers and Technology, 18.07.2019 01:30

Advanced Placement (AP), 18.07.2019 01:30



English, 18.07.2019 01:30



