Answers: 3


Other questions on the subject: Physics

Physics, 22.06.2019 05:00, anime333
Modern physics a photon emitted from an excited hydrogen atom has an energy of 3.02 electron volts. which electron energy-level transition would produce this photon? a. n=1 to n=6 b. n=2 to n=6 c. n=6 to n=1 d. n=6 to n=2 i chose b but the correct answer is d can someone tell me why? and what's the difference?
Answers: 1

Physics, 22.06.2019 05:30, 444x4gang4
Because light travels in a straight line and casts a shadow, isaac newton hypothesized that light is
Answers: 1

Physics, 22.06.2019 07:30, anonymous1813
Some material consisting of a collection of microscopic objects is kept at a high temperature. a photon detector capable of detecting photon energies from infrared through ultraviolet observes photons emitted with energies of 0.3 ev, 0.5 ev, 0.8 ev, 2.0ev, 2.5ev, and 2.8ev. these are the only photon energies observed. (a) draw and label a possible energy-level diagram for one of the microscopic objects, which has four bound states. on the diagram, indicate the transitions corresponding to the emitted photons. explain briefly. (b) would a spring–mass model be a good model for these microscopic objects? why or why not? (c) the material is now cooled down to a very low temperature, and the photon detector stops detecting photon emissions. next, a beam of light with a continuous range of energies from infrared through ultraviolet shines on the material, and the photon detector observes the beam of light after it passes through the material. what photon energies in this beam of light are observed to be significantly reduced in intensity (“dark absorption lines”)? explain briefly.
Answers: 3

Physics, 22.06.2019 15:30, hotsaucerman567
What is a subatomic particle with a negative charge and very little mass?
Answers: 1
You know the right answer?
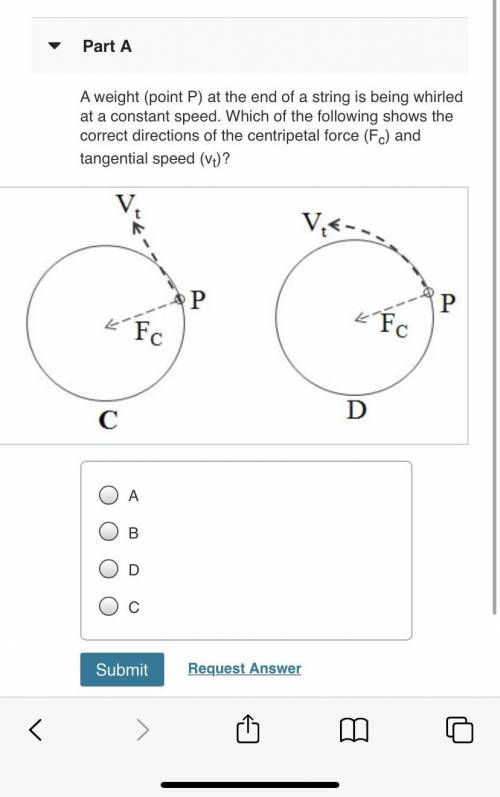
Please Help with this
...
...
Questions in other subjects:

Physics, 30.10.2020 03:40

Mathematics, 30.10.2020 03:40

Mathematics, 30.10.2020 03:40

Mathematics, 30.10.2020 03:40

Mathematics, 30.10.2020 03:40

Mathematics, 30.10.2020 03:40


History, 30.10.2020 03:40

Mathematics, 30.10.2020 03:40

Biology, 30.10.2020 03:40