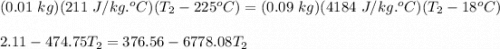Physics, 11.04.2021 14:00 sihamabdalla591
HELP
A student did an experiment to determine the
specific heat capacity of an unknown metal.
She heated 1.00 x 10- kg of the metal to 225°C
and quickly placed it in an insulated container
(negligible specific heat capacity) that contained
0.0900 kg of water at a temperature of 18.0°C.
What is the final temperature of the water if the
specific heat capacity of the metal is
2.11 x 102 J/kg.°C?

Answers: 1


Other questions on the subject: Physics


Physics, 22.06.2019 17:00, StupidFatChipmunk
What equation is used to calculate the weight of an object on a plant ?
Answers: 1

Physics, 22.06.2019 17:40, sosaucyy
Emmy kicks a soccer ball up at an angle of 45° over a level field. she watches the ball's trajectory and notices that it lands, two seconds after being kicked, about 20 m away to the north. assume that air resistance is negligible, and plot the horizontal and vertical components of the ball's velocity as a function of time. consider only the time that the ball is in the air, after being kicked but before landing. take "north" and "up" as the positive ‑ and ‑directions, respectively, and use ≈10 m/s2 for the downward acceleration due to gravity.
Answers: 2

Physics, 22.06.2019 19:20, anaiyamills
Two kilograms of air within a piston–cylinder assembly executes a carnot power cycle with maximum and minimum temperatures of 800 k and 300 k, respectively. the heat transfer to the air during the isothermal expansion is 60 kj. at the end of the isothermal expansion the volume is 0.4 m3. assume the ideal gas model for the air. determine the thermal efficiency, the volume at the beginning of the isothermal expansion, in m3, and the work during the adiabatic expansion, in kj.
Answers: 1
You know the right answer?
HELP
A student did an experiment to determine the
specific heat capacity of an unknown metal....
specific heat capacity of an unknown metal....
Questions in other subjects:


History, 04.08.2019 11:10


History, 04.08.2019 11:10

English, 04.08.2019 11:10




Mathematics, 04.08.2019 11:10

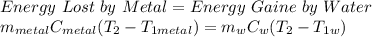
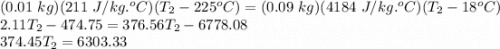 kg (exponent assumed due to missing info in question)
kg (exponent assumed due to missing info in question)