
Physics, 18.01.2021 21:40 CHENDESHEN
How does the density of gas particles inside your tires compare with the density of gas particles in the air outside your tires

Answers: 3


Other questions on the subject: Physics



Physics, 22.06.2019 16:30, sti192
The air in an automobile tire with a volume of 2.60 ft3 is at 70°f and 21 psig. determine the amount of air that must be added to raise the pressure to the recommended value of 30 psig. assume the atmospheric pressure to be 14.6 psia and the temperature and the volume to remain constant. the gas constant of air is ru = 53.34ft⋅lbflbm⋅r(1 psia144 lbf/ft2) = 0.3704 psia⋅ft3lbm⋅r the amount of air that must be added to raise the pressure is lbm
Answers: 3

Physics, 22.06.2019 16:30, lunarwoo
A14 kg rock starting from rest free falls through a distance of 5.0 m with no air resistance. find the momentum change of the rock caused by its fall and the resulting change in the magnitude of earths velocity. earth mass is 6.0 * 10^24 kg. show your work assuming the rock earth system is closed.
Answers: 2
You know the right answer?
How does the density of gas particles inside your tires compare with the density of gas particles in...
Questions in other subjects:

Mathematics, 23.11.2019 06:31




Mathematics, 23.11.2019 06:31

Mathematics, 23.11.2019 06:31

Computers and Technology, 23.11.2019 06:31


Mathematics, 23.11.2019 06:31

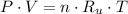 (1)
(1)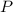 - Pressure.
- Pressure.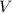 - Volume.
- Volume.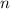 - Molar quantity.
- Molar quantity.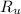 - Ideal gas constant.
- Ideal gas constant.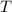 - Temperature.
- Temperature. 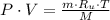
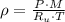 (2)
(2)


