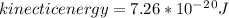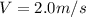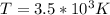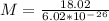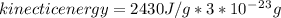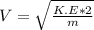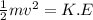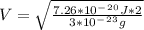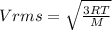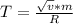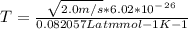The latent heat of vaporization for water at room temperature is 2430 J/g.
1. How much kinetic energy does each water molecule that evaporates possess before it evaporates?
2. Find the pre-evaporation rms speed of a water molecule that is evaporating.
3. What is the effective temperature of these molecules (modeled as if they were already in a thin gas)?
4. Why do these molecules not burn you
a. These molecules got to be slow-moving in collisions that made other molecules fast-moving; the average molecular energy decreases.
b. These molecules got to be slow-moving in collisions that made other molecules fast-moving; the average molecular energy is unaffected.
c. These molecules got to be fast-moving in collisions that made other molecules slow-moving; the average molecular energy is unaffected.
d. These molecules got to be fast-moving in collisions that made other molecules slow-moving; the average molecular energy increases.

Answers: 2


Other questions on the subject: Physics


Physics, 22.06.2019 09:30, ayeheavymetal
The graph represents the distance a car travels over time while on the highway. which statement about the car's trip is accurate? a) the car does not move over time. b) the car travels at a constant velocity. c) the car's velocity increases constantly over time. d) the car's velocity decreases constantly over time.
Answers: 1

Physics, 22.06.2019 10:00, Trinhphuongtran
One object has a mass of 1 kg and another object has a mass of 3 kg. if the speeds are the same, which of the following is true about their kinetic energy?
Answers: 2
You know the right answer?
The latent heat of vaporization for water at room temperature is 2430 J/g.
1. How much kinetic ener...
Questions in other subjects:





History, 19.07.2019 04:50



Mathematics, 19.07.2019 04:50

Physics, 19.07.2019 04:50