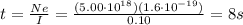Answers: 2


Other questions on the subject: Physics

Physics, 22.06.2019 00:10, oktacos
The energy released by a chemical reaction can be measured using a calorimeter. when barium hydroxide octahydrate crystals are reacted with dry ammonium chloride inside of a coffee cup calorimeter, the temperature of the 18.00 g of water in the calorimeter decreases from 30.0°c to 8.0°c. the equation for calculating energy absorbed or released by a reaction is: where q is the energy released or absorbed, m is the mass of water in the calorimeter, cp is the specific heat of water, and δt is the observed temperature change. if the specific heat of liquid water is 4.19 j/g·°c, how much energy was absorbed by the reaction?
Answers: 3

Physics, 22.06.2019 11:20, puppylove899
Wave functions describe orbitals in a hydrogen atom. each function is characterized by 3 quantum numbers: n, l, and ml. if the value of n = 2: the quantum number l can have values from to . the total number of orbitals possible at the n = 2 energy level is .
Answers: 3


Physics, 22.06.2019 16:50, anaismami1
During a medieval siege of a castle, the attacking army uses a trebuchet to hurl heavy stones at the castle walls. if the trebuchet launches the stones with a velocity of at an angle of 50.0°, (a) how long does it take the stone to hit the ground? (b) what is the maximum distance that the trebuchet can be from the castle wall to be in range? (c) how high will the stones go? show all your work.
Answers: 2
You know the right answer?
If a metal wire carries a current of 0.10 A, how long does it take for 5.00x1018 electrons to pass a...
Questions in other subjects:



Mathematics, 09.04.2021 20:20

Mathematics, 09.04.2021 20:20

Biology, 09.04.2021 20:20




Mathematics, 09.04.2021 20:20

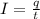
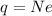
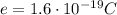 is the fundamental charge (the charge of one electron)
is the fundamental charge (the charge of one electron)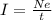
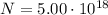 is the number of electrons
is the number of electrons