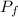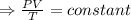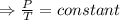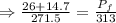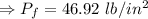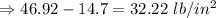Physics, 30.03.2020 21:04 anthonybowie99
An automobile tire having a temperature of −1.5 ◦C (a cold tire on a cold day) is filled to a gauge pressure of 26 lb/in2 . What would be the gauge pressure in the tire when its temperature rises to 40◦C? For simplicity, assume that the volume of the tire remains constant, that the air does not leak out and that the atmospheric pressure remains constant at 14.7 lb/in2 . Answer in units of lb/in2 .

Answers: 3


Other questions on the subject: Physics


Physics, 22.06.2019 06:30, corcoranrobert1959
At very high pressures, gases become and will eventually a) more dense; become hotter b) more dense; change to a liquid or solid c) less dense; combust d) less dense; turn into a liquid
Answers: 1

Physics, 22.06.2019 17:30, funnybugy16
How does the entropy of steam compare to the entropy of ice?
Answers: 2

Physics, 22.06.2019 18:00, sarahidan
Aprisoner is forced to go into one of three rooms, but he can choose which room. the first room is ablaze with fire. the second one is rigged with explosives that will go off as soon as he enters. the third contains a pair of lions who haven't eaten in years. which room should he choose to survive?
Answers: 2
You know the right answer?
An automobile tire having a temperature of −1.5 ◦C (a cold tire on a cold day) is filled to a gauge...
Questions in other subjects:

History, 27.09.2020 18:01

Biology, 27.09.2020 18:01



Biology, 27.09.2020 18:01

Mathematics, 27.09.2020 18:01


English, 27.09.2020 18:01


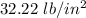
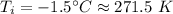
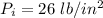
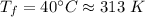 on a hot day then
on a hot day then