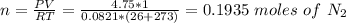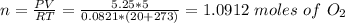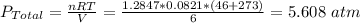A quantity of n2 gas originally held at 4.75 atm pressure in a 1.00-L container at 26c is transerred to a 10.0-L container at 20c. a quanity of o2 gas originally at 5.25 atm and 26c in a 5.00-L container is transferred to this same container. what is the total pressure in the new container

Answers: 2


Other questions on the subject: Physics



You know the right answer?
A quantity of n2 gas originally held at 4.75 atm pressure in a 1.00-L container at 26c is transerred...
Questions in other subjects:


Spanish, 12.12.2020 15:50

Mathematics, 12.12.2020 15:50


Mathematics, 12.12.2020 15:50


Mathematics, 12.12.2020 15:50



English, 12.12.2020 15:50