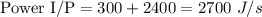Physics, 14.02.2020 03:08 kellynadine02
Steam at a temperature TH = 205 ∘C and p = 1.00 atm enters a heat engine at an unknown flow rate. After passing through the heat engine, it is released at a temperature TC = 100 ∘C and p = 1.00 atm . The measured power output P of the engine is 300 J/s , and the exiting steam has a heat transfer rate of HC = 2400 J/s . Find the efficiency e of the engine and the molar flow rate n/t of steam through the engine. The constant pressure molar heat capacity Cp for steam is37.47 J/(mol⋅K) .
Part A:
What is the efficiency of the heat engine?
Express the efficiency numerically to three significant figures.

Answers: 2


Other questions on the subject: Physics



Physics, 22.06.2019 11:30, tommyaberman
You've already seen the value of 9.8 in this lesson. what's this value called? what quantity does it represent?
Answers: 2

Physics, 22.06.2019 12:30, lanaasad7733
An ice-making machine inside a refrigerator operates in a carnot cycle. it takes heat from liquid water at 0.0 degrees celsius and rejects heat to a room at a temperature of 19.2 degrees celsius. suppose that liquid water with a mass of 76.3kg at 0.0 degrees celsius is converted to ice at the same temperature. take the heat of fusion for water to be l_f = 3.34*10^5 j/kg. how much energy e must be supplied to the device? express your answer in joules.
Answers: 1
You know the right answer?
Steam at a temperature TH = 205 ∘C and p = 1.00 atm enters a heat engine at an unknown flow rate. Af...
Questions in other subjects:



History, 14.11.2020 01:20

History, 14.11.2020 01:20

Mathematics, 14.11.2020 01:20





Biology, 14.11.2020 01:20