
How much heat energy is required to convert 93.4 g of solid ethanol at − 114.5 ° c to gasesous ethanol at 149.8 ° c ? the molar heat of fusion of ethanol is 4.60 kj/mol , and its molar heat of vaporization is 38.56 kj/mol . ethanol has a normal melting point of − 114.5 ° c and a normal boiling point of 78.4 ° c . the specific heat capacity of liquid ethanol is 2.45 j / g ⋅ ° c , and that of gaseous ethanol is 1.43 j / g ⋅ ° c .

Answers: 3


Other questions on the subject: Physics

Physics, 21.06.2019 21:40, lolz55678
Achemistry student needs of 40ml diethylamine for an experiment. by consulting the crc handbook of chemistry and physics, the student discovers that the density of diethylamine is 0.7 . calculate the mass of diethylamine the student should weigh out. round your answer to significant digits.
Answers: 1

Physics, 22.06.2019 00:00, raeganwelch
Ahypothesis is? close agreement by competent observers of observations of the same phenomena. a guess that has been tested over and over again and always found to be true. an educated guess that has yet to be proven by experiment. the long side of a right triangle. a synthesis of a large collection of information that includes guesses.
Answers: 2


Physics, 22.06.2019 12:00, drivinghydra
Aboat radioed a distress call to a coast guard station. at the time of the call, a vector a from the station to the boat had a magnitude of 45.0 km and was directed 15.0° east of north. a vector from the station to the point where the boat was later found is = 30.0 km, 15.0° north of east. what are the components of the vector from the point where the distress call was made to point where the boat was found? in other words, what are the components of vector c = b - a?
Answers: 3
You know the right answer?
How much heat energy is required to convert 93.4 g of solid ethanol at − 114.5 ° c to gasesous ethan...
Questions in other subjects:

Engineering, 25.11.2021 05:30

Computers and Technology, 25.11.2021 05:30

Mathematics, 25.11.2021 05:30




History, 25.11.2021 05:30



Mathematics, 25.11.2021 05:30

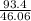 = 2.02 moles
= 2.02 moles

