
(a) it is difficult to extinguish a fire on a crude oil tanker, because each liter of crude oil releases 2.80×107j of energy when burned. to illustrate this difficulty, calculate the number of liters of water that must be expended to absorb the energy released by burning 1.00 l of crude oil, if the water’s temperature rises from 20.0°c to 100°c , it boils, and the resulting steam’s temperature rises to 300°c at constant pressure. (b) discuss additional complications caused by the fact that crude oil is less dense than water.

Answers: 1


Other questions on the subject: Physics

Physics, 22.06.2019 06:30, robertrkumar1
At very high pressures, gases become and will eventually a) more dense; become hotter b) more dense; change to a liquid or solid c) less dense; combust d) less dense; turn into a liquid
Answers: 2

Physics, 22.06.2019 23:20, libbybacon7
Consider a production line with five stations. station 1 can produce a unit in 9 minutes. station 2 can produce a unit in 10 minutes. station 3 has two identical machines, each of which can process a unit in 12 minutes (each unit only needs to be processed on one of the two machines. station 4 can produce a unit in 5 minutes. station 5 can produce a unit in 8 minutes. which station is the bottleneck station?
Answers: 2

Physics, 23.06.2019 01:10, jacquilynfoster7
Calculate 9 ∙ 10-5 divided by 3 ∙ 10-9. (box after the "10" in answer is for the
Answers: 1
You know the right answer?
(a) it is difficult to extinguish a fire on a crude oil tanker, because each liter of crude oil rele...
Questions in other subjects:

Computers and Technology, 05.03.2021 17:20


Mathematics, 05.03.2021 17:20


Chemistry, 05.03.2021 17:20

Social Studies, 05.03.2021 17:20


Physics, 05.03.2021 17:20


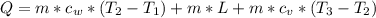
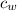 = specific heat capacity of water = 4184 J/(kg*K)
= specific heat capacity of water = 4184 J/(kg*K)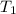 = 20 + 273.15 = 293.15 K
= 20 + 273.15 = 293.15 K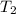 = 100 + 273.15 = 393.15 K
= 100 + 273.15 = 393.15 K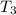 = 300 + 273.15 = 573.15 K
= 300 + 273.15 = 573.15 K 

