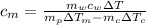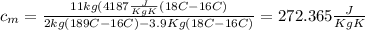Calculate the specific heat of a metal from the following data. a container made of the metal has a mass of 3.9 kg and contains 11 kg of water. a 2.0 kg piece of the metal initially at a temperature of 189°c is dropped into the water. the container and water initially have a temperature of 16.0°c, and the final temperature of the entire system is 18.0°c.

Answers: 3


Other questions on the subject: Physics

Physics, 21.06.2019 22:00, jby
Atank holds 1000 gallons of water, which drains from the bottom of the tank in half an hour. the values in the table show the volume v of water remaining in the tank (in gallons) after t minutes. t(min) 5 10 15 20 25 30v(gal) 694 444 250 111 28 0(a) if p is the point (15, 250) on the graph of v, find the slopes of the secant lines pq when q is the point on the graph with t = 5, 10, 20, 25, and 30.(b) estimate the slope of the tangent line at p by averaging the slopes of two secant lines.(c) use a graph of the function to estimate the slope of the tangent line at p. (this slope represents the rate at which the water is flowing from the tank after 15 minutes.)show all steps.
Answers: 2


Physics, 22.06.2019 10:00, amyaacrawford86
Because air contracts as it cools, the air pressure inside a freezer is typically lower than on the outside. why do ice cubes inside a freezer tend to shrink over time? a. the ice dissolves oxygen from the air, forming a denser crystalline matrix. b. the ice reacts chemically with carbon dioxide in the air, forming gaseous carbon compounds. c. the ice melts, and then the liquid freezes as ice crystals on the bottom of the freezer. d. the ice sublimes, and then the water vapor deposits as ice crystals on the sides of the freezer.
Answers: 1
You know the right answer?
Calculate the specific heat of a metal from the following data. a container made of the metal has a...
Questions in other subjects:

Mathematics, 14.04.2020 22:18

English, 14.04.2020 22:18

Mathematics, 14.04.2020 22:18







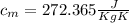
 = mass of the container = 3.9 kg
= mass of the container = 3.9 kg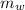 = mass of the water inside of the container= 11 kg
= mass of the water inside of the container= 11 kg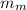 =mass of the metal= 2 kg
=mass of the metal= 2 kg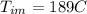 initital temperature of the metal
initital temperature of the metal initital temperature of the water
initital temperature of the water initital temperature of the container
initital temperature of the container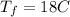 final equilibrium temperature
final equilibrium temperature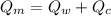
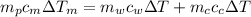
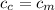
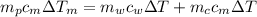
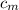 from the last expression we got:
from the last expression we got: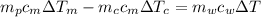
![c_m [m_p \Delta T_m -m_c \Delta T_c] =m_w c_w \Delta T](/tpl/images/0387/5562/73942.png)