Xenon
Explanation:
The average atomic mass of an element is defined as the sum of the masses of its isotopes,each multiplied by their natural abundances.
Option A:
Argon has a average atomic mass of 
Option B:
Xenon has a average atomic mass of 
Option C:
Krypton has a average atomic mass of 
Option D:
Neon has a average atomic mass of 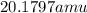
So,Xenon has the highest average atomic mass.
















