Physics, 20.11.2019 22:31 lilquongohard
The molar enthalpy of fusion (δh°) of bromine is 10.570 kj/mol, and the molar entropy of fusion (δs°) of bromine is 39.750 j/mol k. using this information, estimate the freezing point of bromine. (you must answer in kelvin.)

Answers: 3


Other questions on the subject: Physics

Physics, 21.06.2019 20:30, LunaSpellman
Which statement best describes how private industry decides where to spend the money on research and development
Answers: 1

Physics, 22.06.2019 12:10, davidoj13
Awater slide of length l has a vertical drop of h. abby's mass is m. an average friction force of magnitude f opposes her motion. she starts down the slide at initial speed vi. use work-energy ideas to develop an expression for her speed at the bottom of the slide. then evaluate your result using unit analysis and limiting case analysis. express your answer in terms of the variables h, m, l, vi, f and appropriate constants. vf v f
Answers: 2

Physics, 22.06.2019 17:20, Queenhagar
In a system with only a single force acting upon a body, what is the relationship between the change in kinetic energy and the work done by the force? answers: work is equal to the change in kinetic energy. work depends on the square of the change in potential energy. work is equal to the negative of the change in kinetic energy. work is equal to the square of the change in kinetic energy
Answers: 2
You know the right answer?
The molar enthalpy of fusion (δh°) of bromine is 10.570 kj/mol, and the molar entropy of fusion (δs°...
Questions in other subjects:




SAT, 20.09.2020 14:01


Mathematics, 20.09.2020 14:01




 = Molar enthalpy of fusion = 10.57 kJ/mol
= Molar enthalpy of fusion = 10.57 kJ/mol = Molar entropy of fusion = 39.75 J/mol
= Molar entropy of fusion = 39.75 J/mol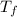 = Freezing point
= Freezing point