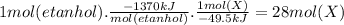Physics, 11.11.2019 22:31 adiboo2004
Assume that the complete combustion of one mole of ethanol to carbon dioxide and water liberates 1370 kj of energy (δ°′=−1370 kj/mol ). if the energy generated by the combustion of ethanol is entirely converted to the synthesis of a hypothetical compound x, calculate the number of moles of the compound that could theoretically be generated. use the value δ°′compound x=−45.9 kj/mol . round your answer to two significant figures.

Answers: 1


Other questions on the subject: Physics


Physics, 22.06.2019 19:40, rileybaby34
Uranium has two naturally occurring isotopes. 238u has a natural abundance of 99.3% and 235u has an abundance of 0.7%. it is the rarer 235u that is needed for nuclear reactors. the isotopes are separated by forming uranium hexafluoride uf6, which is a gas, then allowing it to diffuse through a series of porous membranes. 235uf6 has a slightly larger rms speed than 238uf6 and diffuses slightly faster. many repetitions of this procedure gradually separate the two isotopes. what is the ratio of the rms speed of 235uf6 to that of 238uf6? express your answer to five significant figures.
Answers: 3

Physics, 22.06.2019 23:00, Miguel1310
According to the image below, which season is about to begin in the southern hemisphere? a. summer b. autumn c. winter d. spring
Answers: 1

Physics, 23.06.2019 01:00, kaziyahf2006
When solid surfaces slide over each other the kind of friction that occurs is called friction
Answers: 2
You know the right answer?
Assume that the complete combustion of one mole of ethanol to carbon dioxide and water liberates 137...
Questions in other subjects:


Chemistry, 23.09.2021 02:10

Mathematics, 23.09.2021 02:10






Business, 23.09.2021 02:10