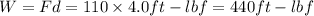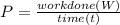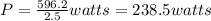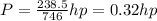Answers: 1


Other questions on the subject: Physics

Physics, 22.06.2019 00:10, oktacos
The energy released by a chemical reaction can be measured using a calorimeter. when barium hydroxide octahydrate crystals are reacted with dry ammonium chloride inside of a coffee cup calorimeter, the temperature of the 18.00 g of water in the calorimeter decreases from 30.0°c to 8.0°c. the equation for calculating energy absorbed or released by a reaction is: where q is the energy released or absorbed, m is the mass of water in the calorimeter, cp is the specific heat of water, and δt is the observed temperature change. if the specific heat of liquid water is 4.19 j/g·°c, how much energy was absorbed by the reaction?
Answers: 3



Physics, 23.06.2019 07:30, linacelina6027
For two variables in a direct proportion, what is the result of doubling one variable?
Answers: 1
You know the right answer?
A110-pound person pulls herself up 4.0 feet. this took her 2.5 seconds. how much power was developed...
Questions in other subjects:


Mathematics, 05.05.2020 11:45

History, 05.05.2020 11:45

Mathematics, 05.05.2020 11:45

English, 05.05.2020 11:45




English, 05.05.2020 11:45